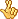To try to answer that question, a team of German bioengineers surgically installed coiled strips of optical fibers in the ears of deaf gerbils. While they still had their hearing, the gerbils had learned to hurdle a small barrier upon hearing an alarm. Now researchers sent a pulse of blue laser light deep into the animals ears. They jumped. he experiment was part of a
study published Wednesday seeking to improve upon cochlear implants electronic devices that stimulate auditory neurons to partially restore hearing. Instead of using electrical currents, scientists are trying to determine whether optogenetics, a new field that uses light to control living cells, could one day help improve someones sense of hearing.
Although it could take decades to use optogenetics-based technologies in humans, researchers are beginning to demonstrate that light, not electricity, may be the best way to convey the rich information contained in sound. Through my experience with patients, Ive recognized the huge potential of cochlear implants, said Tobias Moser, director of the Institute for Auditory Neuroscience at the University Medical Center Göttingen and lead author of the new study. At the same time Ive also witnessed the shortcomings. Patients with cochlear implants often describe the sound quality through the devices as harsh and tinny. Listening to music or picking out one voice from several others is often impossible. The goal of the new research, said Moser, is to improve the technology and create a more natural hearing so that patients can recognize the melody in music and speech.
At the most basic level, the act of hearing is transforming sound into electrochemical signals, the language of neurons, that the brain can then interpret. Much of this process occurs in the cochlea, a snail-shaped organ within the inner ear lined with specialized sensory cells called hair cells. When hair cells detect vibration through thin protrusions on their surface, they generate electrical current in neighboring nerve cells that travel to the brain. In people who have dysfunctional or dead hair cells, cochlear implants work by electrically stimulating auditory nerve cells directly. According to Dan Polley, an associate professor at Harvard Medical School and director of the Lauer Tinnitus Research Center who was not involved in the study, the successes and limitations of the cochlear implant are determined by the anatomy of the inner ear.
Within the cochlea, hair cells rest on an organic platform known as the basilar membrane that is floppy and wide on one end and narrow and taut on the other. This biomechanical organization causes hair cells to wiggle in response to specific sound frequencies or pitches that are mapped out smoothly from low to high, like keys on a piano. When implanting cochlear implants, Polley explained, surgeons thread electrodes into specific locations along the basilar membrane to target nerves that are sensitive to particular frequencies. With cochlear implants, however, electrical current spreads itself over a large area and activates not only the targeted nerves but also neighboring cells as well. This impercision distorts and muffles sound.
MORE


 The Science, Health, and Technology Room
The Science, Health, and Technology Room


 Reply With Quote
Reply With Quote